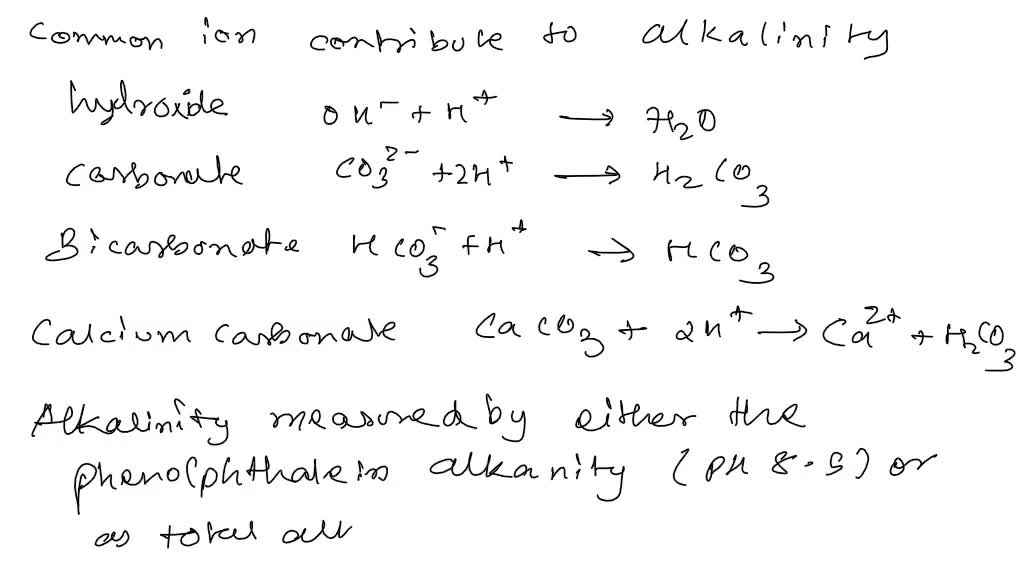SOLVED: Calculate the total alkalinity for a sample of river water whose phenolphthalein alkalinity is known to be 3.0 x 10-5 M, whose pH = 10.0, and whose bicarbonate ion concentration is 1.0 x 10-4 M

SOLVED: Total hardness, carbonate hardness, and noncarbonate hardness. Calculate the alkalinity for the following water in mg/L as CaCO3: Anions: HCO3- 135 mg/L SO4 134 mg/L Cl 0 mg/L Cations: Ca2+ 94