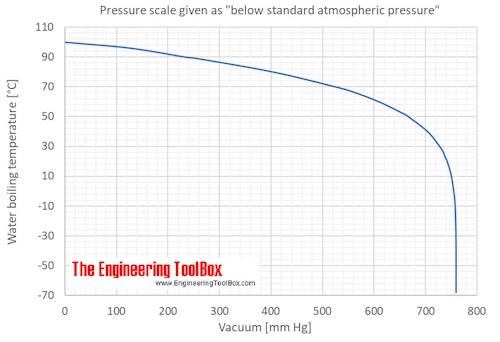
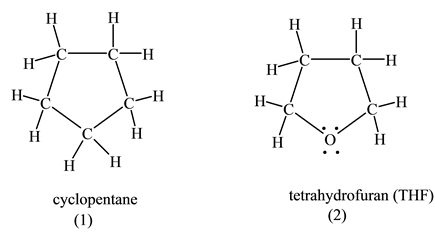a I0 −1 vs T −1 of the THF-water mixture at various THF contents and b... | Download Scientific Diagram

Eutectic and melting points for the four isotopic THF- water mixtures.... | Download Scientific Diagram
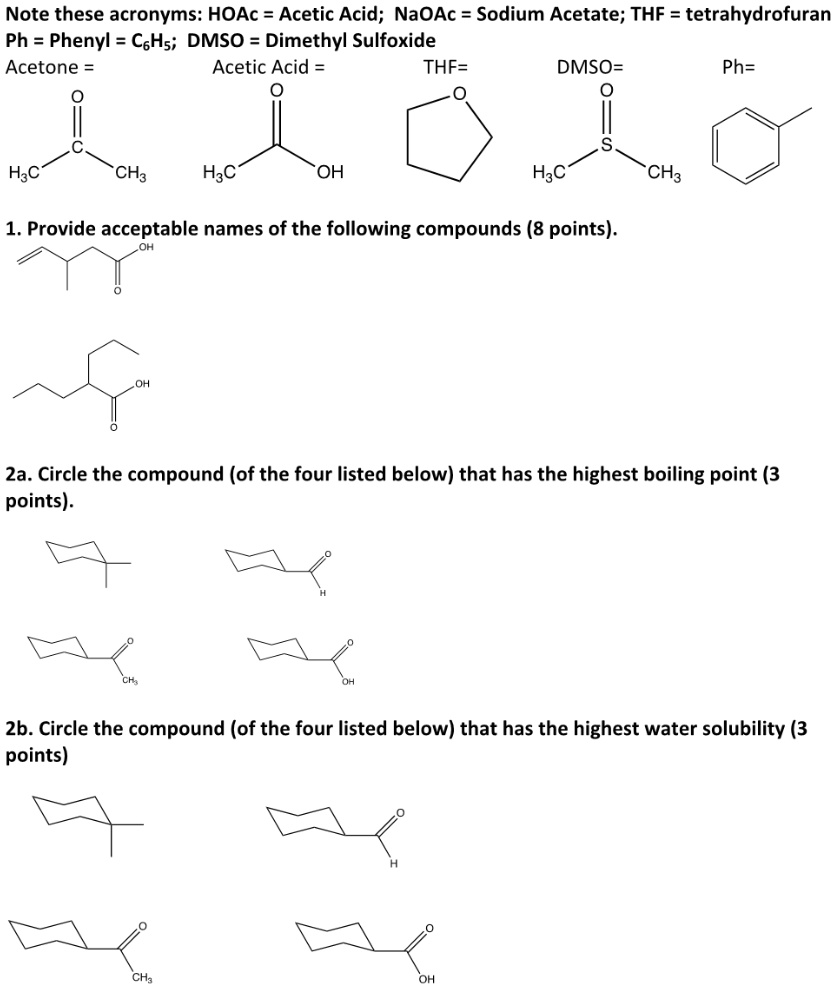
SOLVED: Note these acronyms: HOAc = Acetic Acid; NaOAc = Sodium Acetate; THF = tetrahydrofuran; Ph = Phenyl = C6H5; DMSO = Dimethyl Sulfoxide; Acetone; Acetic Acid; THF; DMSO; Ph. H3C OH
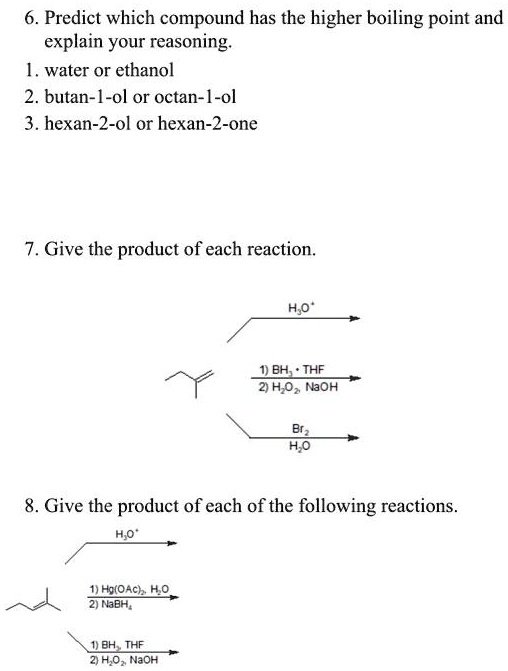
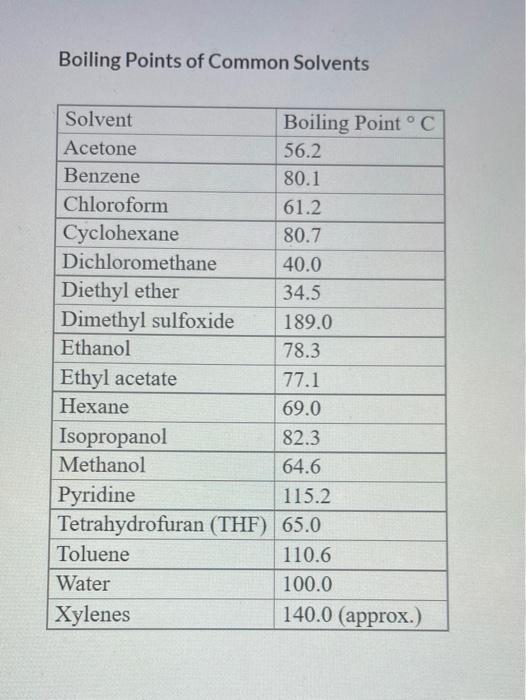
SOLVED: 6. Predict which compound has the higher boiling point and explain your reasoning: water or ethanol. 2. butanol or octanol. 3. hexan-2-ol or hexan-2-one. 7. Give the product of each reaction.

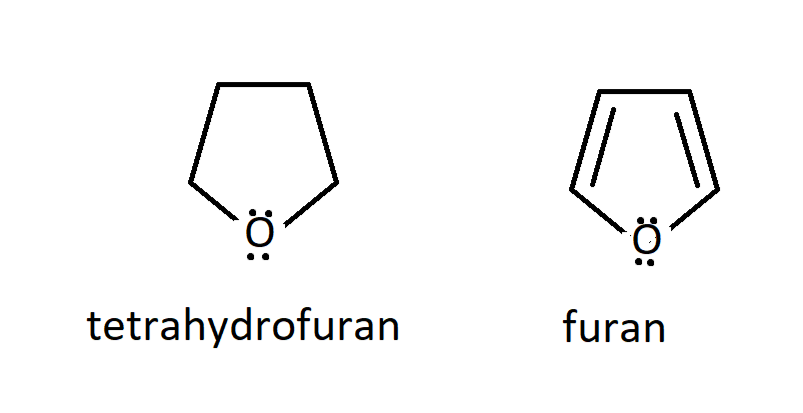
Explain why tetrahydrofuran has a higher boiling point and is much more water soluble than furan, even though both compounds are cyclic ethers containing four carbons. | Homework.Study.com
![High Boiling Point Cyclic Ether | [Common Chemicals & Lab Tools]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation High Boiling Point Cyclic Ether | [Common Chemicals & Lab Tools]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/us/category/images/00277_img02.png)
High Boiling Point Cyclic Ether | [Common Chemicals & Lab Tools]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation