%20nitrate%20in%20a%20boiling%20tube,%20black%20copper%20oxide,%20O2%20and%20a%20brown%20gas%20X%20is%20form.jpg)
On heating blue coloured powder of copper (II) nitrate in a boiling tube, black copper oxide, O2 and a brown gas X is formed
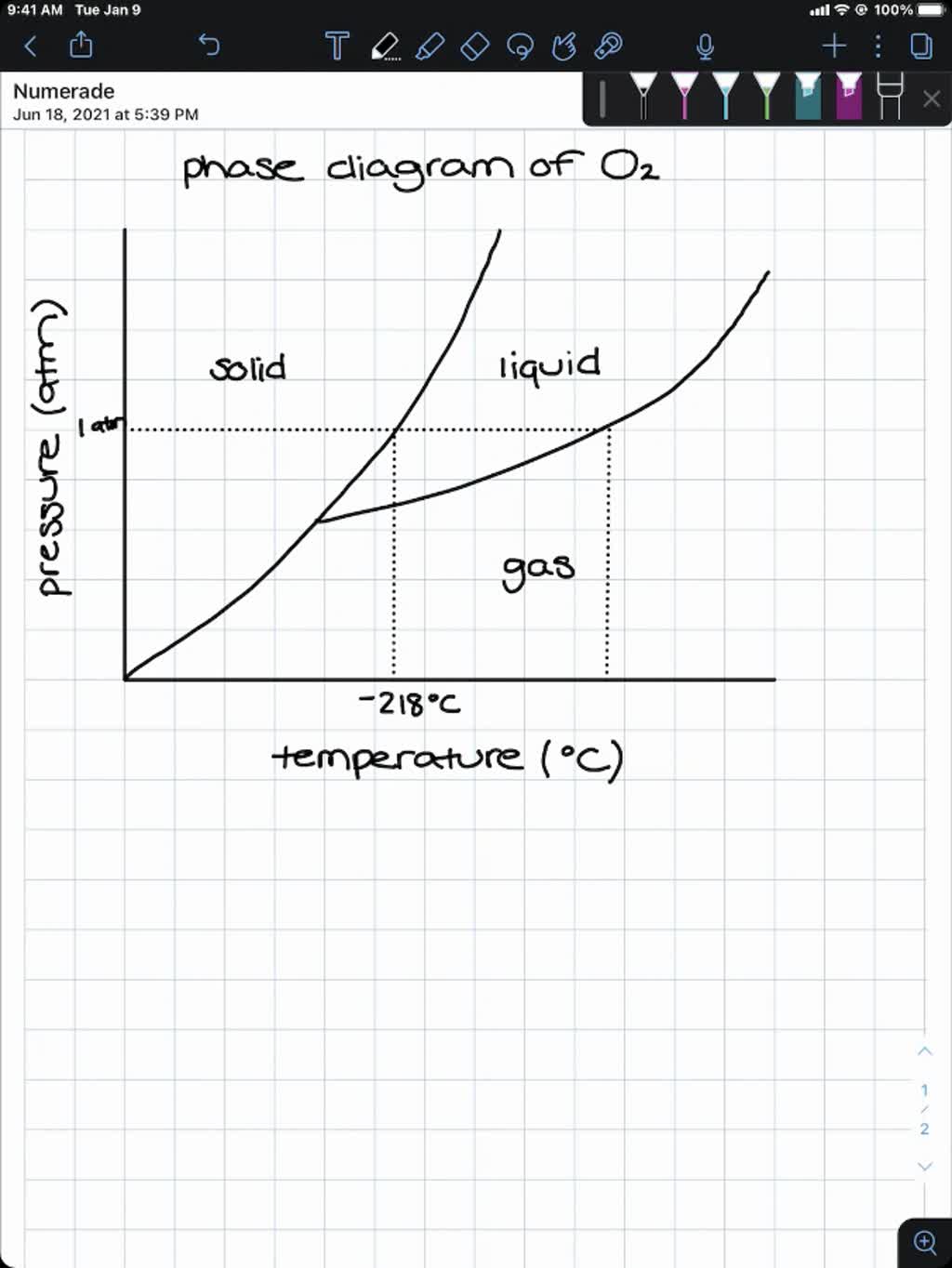
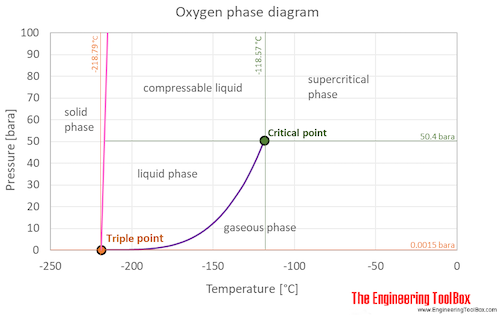
SOLVED:Use graph paper and sketch the phase diagram of oxygen, O2, from the following information: normal melting point, -218^∘ C; normal boiling point, -183^∘ C; triple point, -219^∘ C, 1.10 mmHg; critical

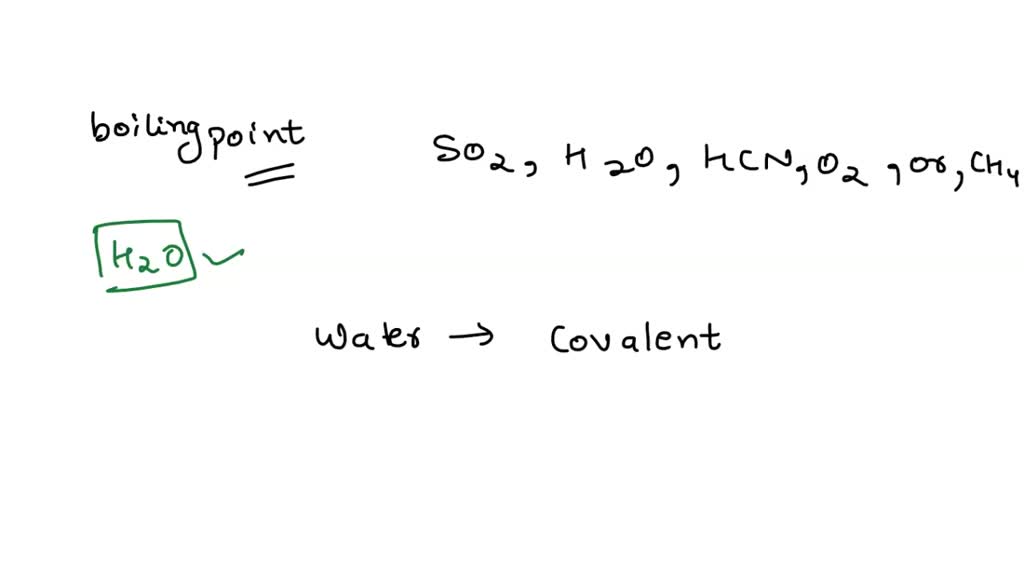
The correct order of normal boiling points of O2, N2, NH3 and CH4 for whom the values of vander waals constant ' a ' are 1.360, 1.390, 4.170 and 2.253 L ^2 atm. mol ^-2 respectively is:

Sketch the phase diagram of oxygen, O2, from the following information: normal melting point, -218 degrees C; normal boiling point, -183 degrees C; triple point, -219 degrees C, 1.10 mmHg; critical point, -