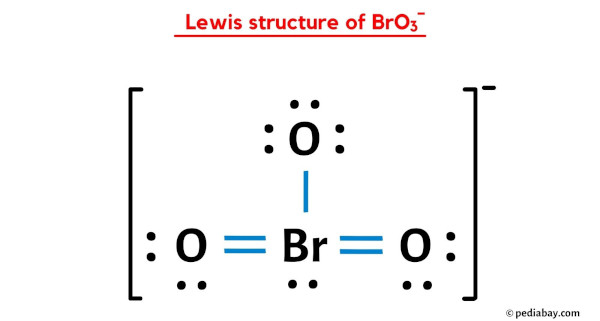
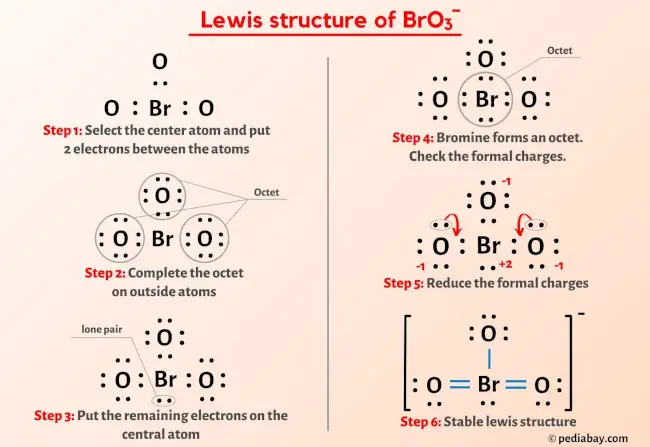
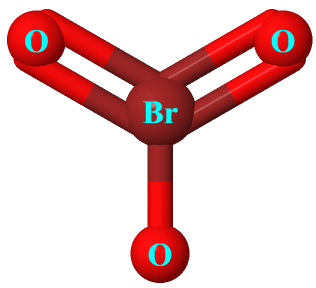
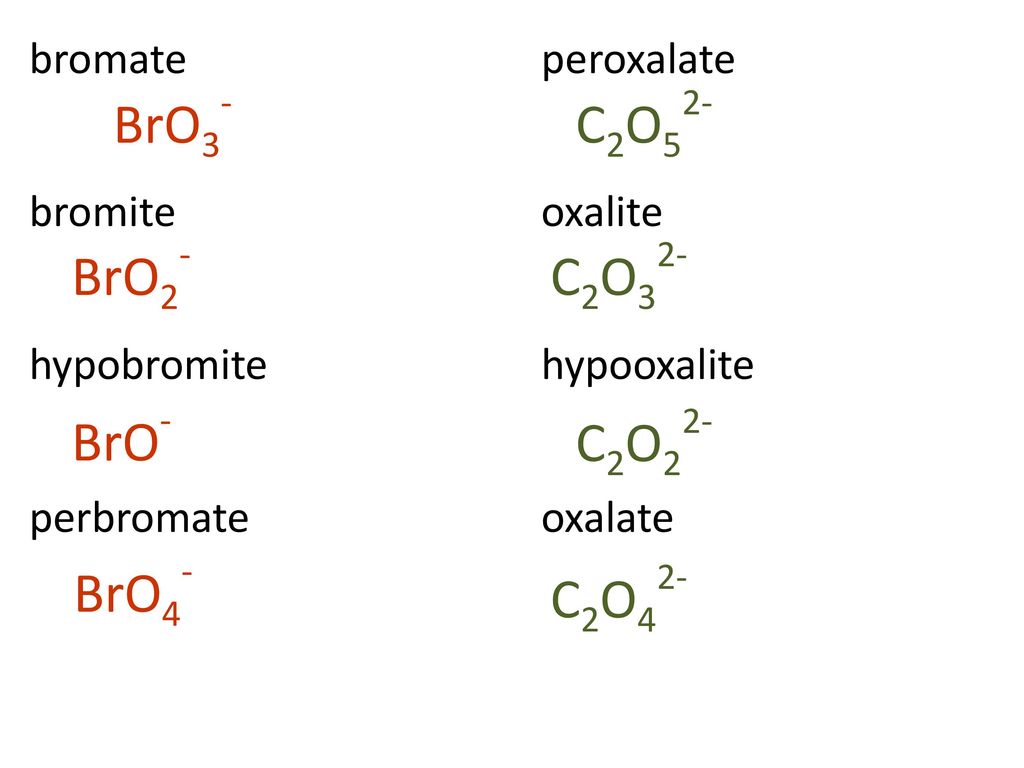
Draw a Lewis structure for BrO3- that obeys the octet rule. Assign oxidation numbers to each atom. | Homework.Study.com

Draw the Lewis structure for the bromate ion BrO3- obeying the octet rule and then draw it when it breaks the octet rule but minimizes formal charges. | Homework.Study.com