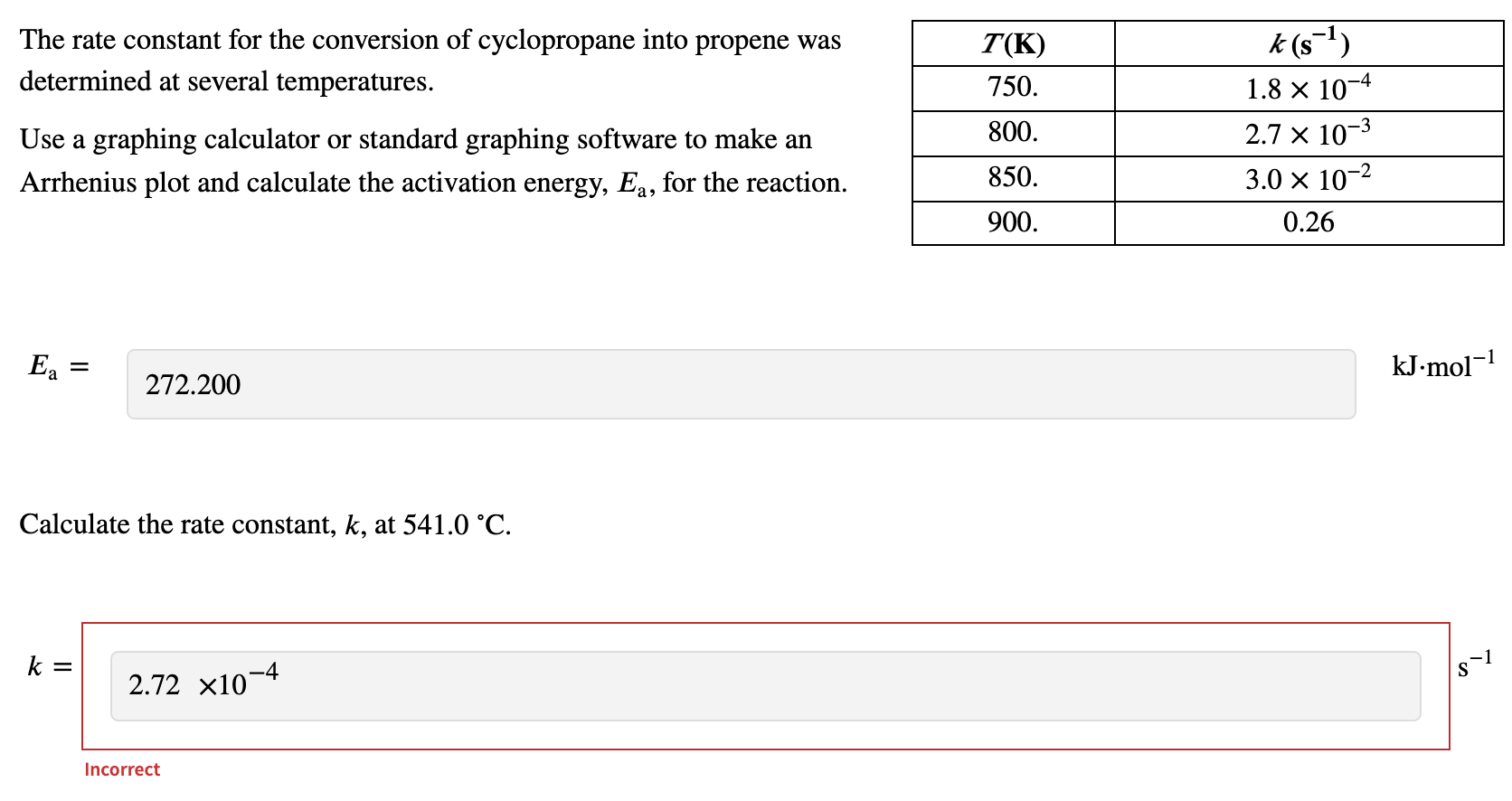
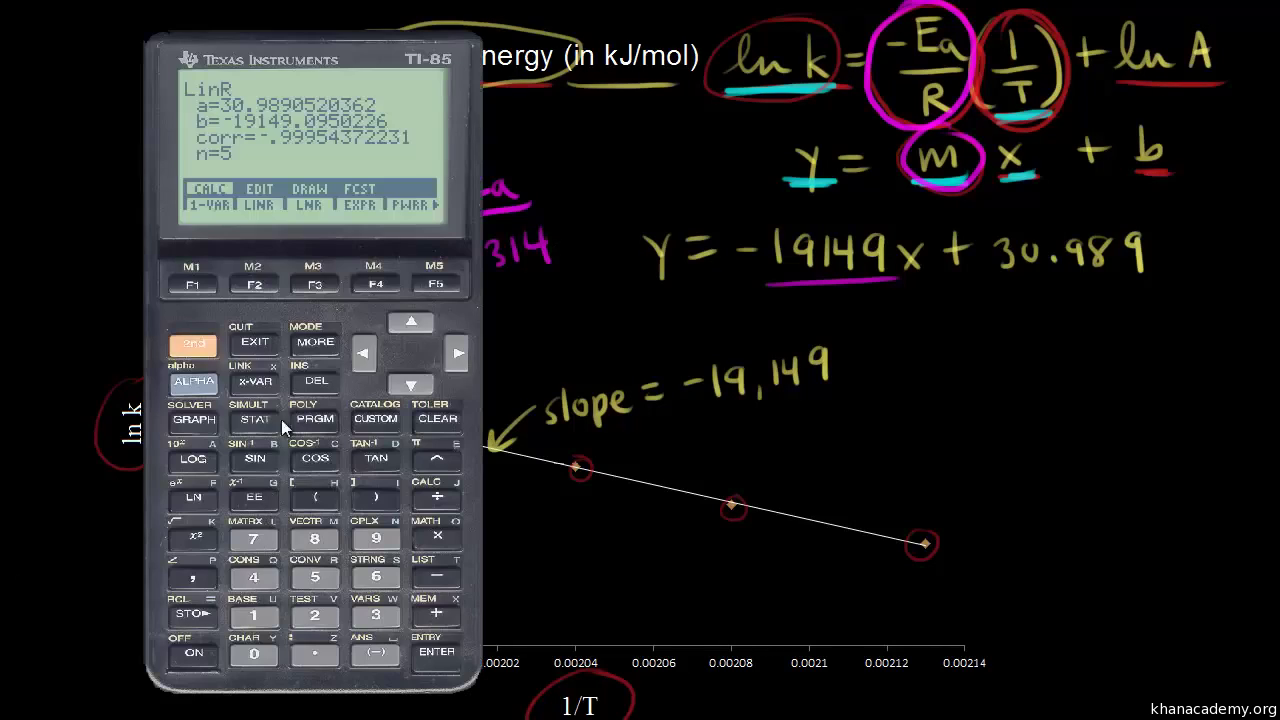
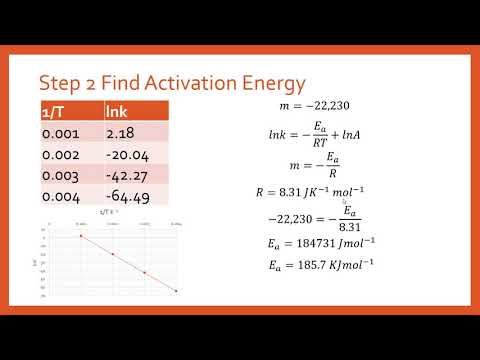
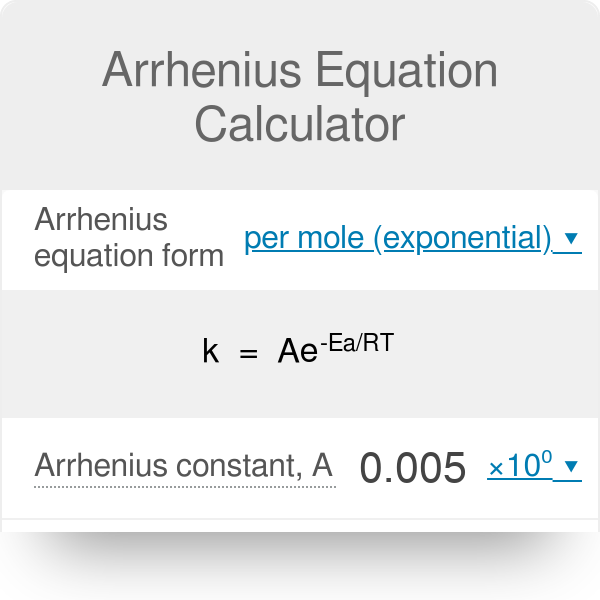
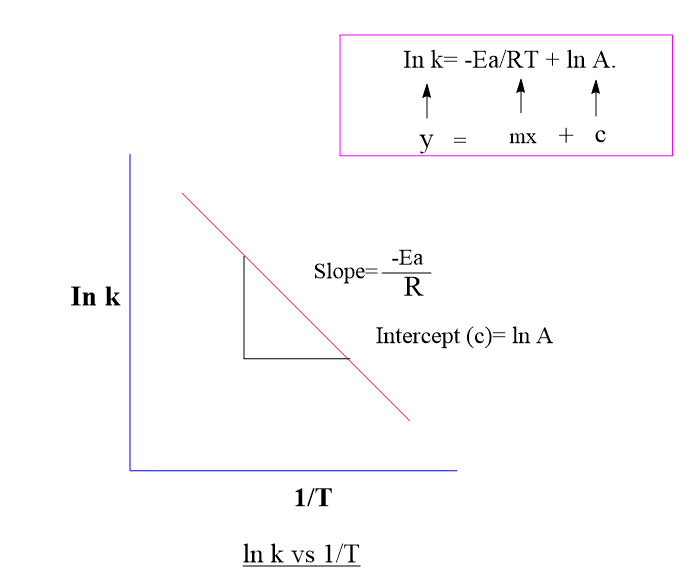
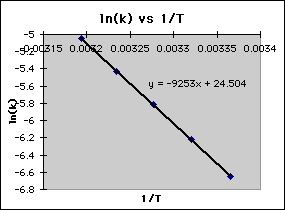
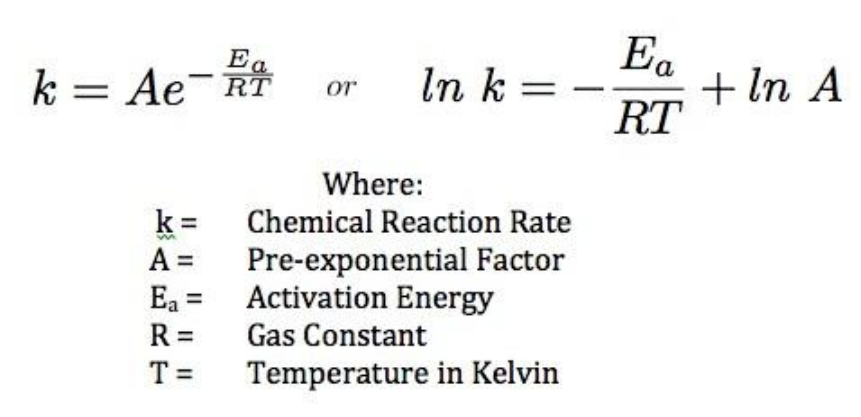Calculate Activation Energy for a Reaction of Which Rate Constant Becomes Four Times When Temperature Changes from 30 °C to 50 °C - Chemistry | Shaalaa.com

Apparent activation energy calculation with Ph 2 CO mixed with C 3 H 8... | Download Scientific Diagram

Activation Energy Calculator - Free Online Tool | How to find Activation Energy? - physicsCalculatorPro.com

Arrhenius & Activation Energy (5.5.9) | Edexcel A Level Chemistry Revision Notes 2017 | Save My Exams

SOLVED: Ok, so I understand the process of doing this, but I'm having trouble inputting it into my calculator. The reaction CH3CHO(g) â†' CH4(g) + CO(g) has a rate constant of 0.213